Starting a fire is a fundamental survival skill, but treating it as a science experiment rather than a matter of luck is the key to consistent success. Many outdoor enthusiasts struggle to create a flame under pressure because they focus on the motions without understanding the underlying principles of combustion. This guide moves beyond simple tutorials to explain the “why” behind the “how.” By exploring fire starting science, we break down the chemistry and physics that govern ignition and burn efficiency. You’ll learn how to control heat, fuel, and oxygen using proven, science-backed techniques to create fire reliably in any environment.
Your Scientific Blueprint: The Fire Triangle Explained
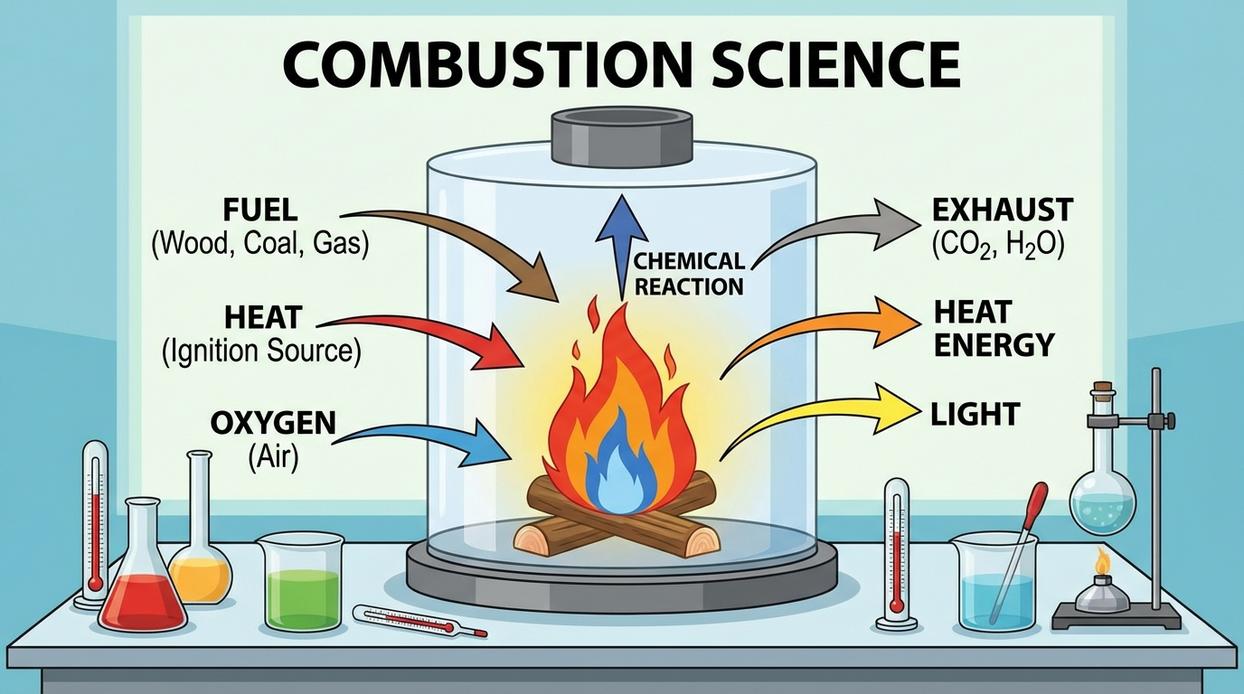
At its core, fire is a rapid chemical reaction called combustion. In simpler terms, it’s chemistry moving fast—and looking dramatic while doing it (like a science experiment that refuses to stay in the beaker). For combustion to happen, three elements must work together: heat, fuel, and an oxidizing agent—usually oxygen.
First, heat. This is the activation energy—the spark, friction, or flame that raises material to its ignition temperature. No heat? No party. Next comes fuel, meaning any combustible material. However, not all fuel is equal. You start with tinder—low ignition temperature and high surface area—then graduate to kindling, and finally larger logs. Think of it as assembling a tiny wooden ladder to success.
Meanwhile, oxygen acts as the catalyst. It reacts with heated fuel to sustain combustion. That’s why fire lays like teepees or log cabins are designed for airflow. A fire needs to breathe (yes, your campfire does yoga).
Understanding this triangle shifts your mindset. You’re not “trying to light a fire.” You’re managing a chemical reaction using fire starting science. If it fails, one element is missing. Always. Check the triangle—and try not to blame the matches.
Method 1: Friction and the Conversion of Motion to Heat
Let’s be honest—few outdoor skills are as frustrating as trying to start a friction fire for the first time. You sweat, your arms burn, and nothing happens except a sad pile of lukewarm dust. Meanwhile, every survival show makes it look effortless (of course it does).
At its core, fire starting science is simple: The principle of friction fire is converting kinetic energy (motion) into thermal energy (heat). Rub two pieces of wood together fast enough, and you create concentrated heat. Eventually, that heat raises fine wood dust to its ignition temperature, forming a glowing ember. Simple in theory. Brutal in practice.
The Bow Drill Technique
This is where mechanical advantage saves your sanity. Instead of spinning a stick between your palms until you blister, a bow rapidly rotates the spindle against a fireboard. As a result, friction concentrates on one small point. The carved notch collects hot dust while allowing oxygen to flow—because without air, your ember suffocates before it’s born.
Wood choice is another common pain point. Resinous wood gums up. Too soft, and you get mush. Too hard, and you polish instead of produce dust. Ideally, pair a harder spindle with a slightly softer, dry, non-resinous fireboard.
One more thing: that V-shaped notch isn’t decorative. It’s an oxygen channel. Keep it clean, angled, and clear—or prepare for another 20 minutes of spinning with nothing to show for it.
Method 2: Optics and Concentrating Solar Energy
If you’ve ever used a magnifying glass to burn a leaf, you’ve already experimented with fire starting science. This method relies on basic optics: a convex lens bends (or refracts) parallel sunlight into a single concentrated point called the focal point. At that precise spot, light energy turns into intense heat—often hot enough to ignite dry tinder.
So how do you make it work reliably in the wild? Let’s break it down.
-
Position the Lens Correctly
Hold your magnifying glass toward the sun and adjust the distance between the lens and your tinder. You’re aiming for the smallest, brightest dot possible. The tighter the light cluster, the higher the temperature. -
Choose the Right Tinder
Dark, fluffy materials work best. Dark colors absorb more radiation instead of reflecting it, while fluffy fibers increase surface area, helping the material reach ignition temperature faster. -
Improvise When Needed
No magnifying glass? Try a clear water-filled plastic bag, a smooth ice lens, or even the polished bottom of a soda can. The principle stays the same—focus sunlight to a sharp focal point.
However, some argue this method is unreliable because it depends on direct sunlight. That’s fair. Cloud cover or late-day angles reduce effectiveness. Still, in clear midday conditions, it’s lightweight, fuel-free, and surprisingly efficient.
Pro tip: steady your hands by bracing your elbows against your knees.
And remember, understanding basics like sunlight ignition complements broader skills such as managing gear weight—especially when you’re focused on decoding backpack weight base weight vs total pack load.
Percussion, Oxidation, and Pyrophoricity in Action
At the heart of this method is pyrophoricity—the ability of a material to ignite when exposed to air. In simple terms, when high‑carbon steel strikes flint or chert, tiny iron fragments shear away. These particles oxidize instantly, producing sparks that can exceed 800°F.
Here’s where positioning matters.
Steel vs. Ferro Rod: A traditional steel striker produces cooler, shorter‑lived sparks. A ferrocerium rod (a synthetic alloy) throws hotter, more dramatic sparks. Think subtle campfire ember vs. Fourth of July sparkler. Both work—but they demand different tinder strategies.
Those steel sparks rely on rapid oxidation—fast rusting, essentially—to glow. Because they’re brief, they need char cloth, which is fabric that has been pyrolyzed (heated without oxygen) to lower its ignition point. It catches a spark and smolders instead of flaming out.
In fire starting science, surface area is everything. More exposed iron means faster oxidation—and better odds of ignition. Pro tip: Pair cooler sparks with ultra‑fine tinder for reliability.
Step Into the Wild With Certainty
You set out to understand what truly makes a flame catch and endure. Now you know that fire starting science is about balancing heat, fuel, and oxygen with intention—not luck. The struggle of a dying spark or smoke without flame comes from imbalance, and that frustration can cost you comfort, safety, and confidence in the wild.
Don’t leave something this critical to chance. Equip yourself with proven techniques, reliable tools, and field-tested guidance designed to eliminate guesswork. Join thousands of outdoor enthusiasts who trust our top-rated wilderness insights—explore our expert resources today and turn every spark into certainty.
